 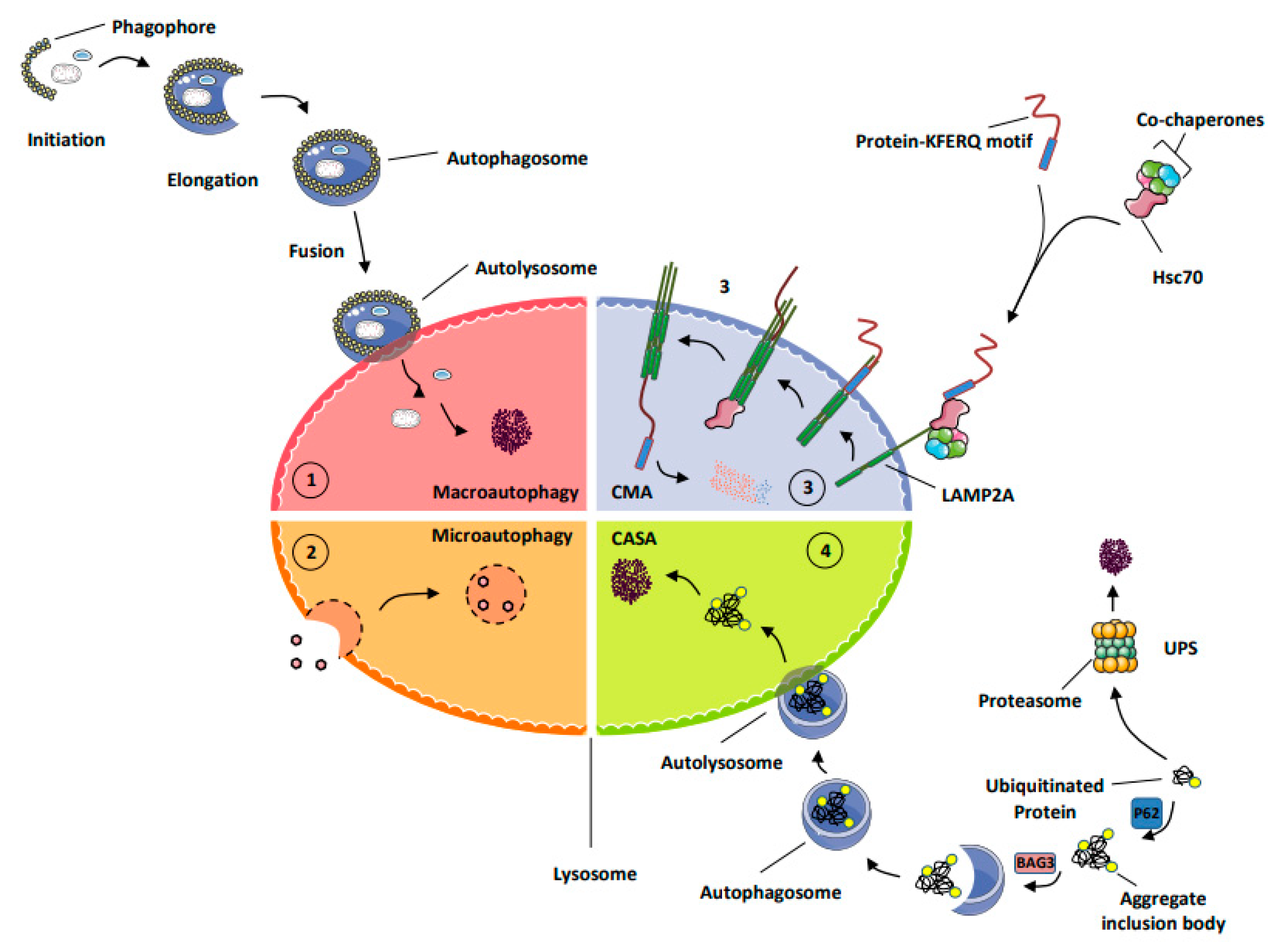
Unfortunately, despite the great efforts of the researchers, these mechanisms are not completely understood. Despite the similarities between these diseases, prions remain unique since epidemiological data support their ability to transmit under natural and experimental conditions between individuals and, to a certain extent, between species ( Mays and Soto, 2016).įor years, TSE research field has focused on characterizing the underlying molecular mechanisms of the basic pathological processes involved in prion pathogenesis and neurodegeneration. Hence, prion diseases share profound similarities with other neurodegenerative disorders associated with the accumulation of misfolded protein aggregates like Alzheimer’s disease, Parkinson’s disease and Huntington’s disease ( Soto, 2003). Accumulation of PrP Sc in the central nervous system (CNS), which is believed to be the main pathogenic event responsible for the pathological changes produced in TSE patients, induces spongiform degeneration, glial activation and neuronal loss ( Wells and McGill, 1992 Wood et al., 1997). According to the protein-only hypothesis ( Prusiner, 1982), TSE are caused by the conformational conversion of native cellular prion protein (PrP c), which is encoded by the prion protein ( PRNP) gene, into an infectious misfolded isoform named scrapie-associated prion protein (PrP Sc). Prion diseases are characterized by a long asymptomatic incubation period and a rapidly progressing pathology that leads inevitably to death. Ovine scrapie was the first TSE described and constitutes one of the most widely studied models of these pathologies ( Pattison and Jones, 1967). In animals, they include, but are not limited to, bovine spongiform encephalopathy (BSE) in cattle, classical and atypical scrapie in sheep and goats, and chronic wasting disease in cervids ( Imran and Mahmood, 2011b). Human prion diseases include kuru, the various forms of Creutzfeldt-Jakob disease (CJD), Gerstmann-Sträussler-Scheinker (GSS) syndrome, fatal familial insomnia, sporadic fatal insomnia, and the variably protease-sensitive prionopathy ( Imran and Mahmood, 2011a). Transmissible spongiform encephalopathies (TSE), or prion diseases, are a group of fatal neurodegenerative disorders that affect both humans and animals ( Prusiner, 1982). In this review, we summarize previous and recent findings concerning the role of autophagy in TSE. Since autophagy is one of the pathways of PrP Sc degradation, and drug-induced stimulation of autophagic flux (the dynamic process of autophagic degradation activity) produces anti-prion effects, new treatments based on its activation have been tested to develop therapeutic strategies for prion diseases. Additionally, autophagy is now emerging as a host defense response in controlling prion infection that plays a protective role by facilitating the clearance of aggregation-prone proteins accumulated within neurons. Altogether, these findings suggest that autophagy is implicated in prion neuropathology and points to an impairment or failure of the process, potentially contributing to the pathogenesis of the disease. It has been suggested that PrP c could play a crucial role in modulating the autophagic pathway in neuronal cells, and the presence of abnormal autophagic activity has been frequently observed in several models of TSE both in vitro and in vivo, as well as in human prion diseases. During the last years, researchers have used a wide range of morphological, genetic and biochemical methods to monitor and manipulate the autophagic pathway and thus determine the specific role of this process in TSE. Thus, the biological role of autophagy in these diseases is still open to debate. The precise connection of this process with prion-related neuropathology, or even whether autophagy is completely beneficial or pathogenic during neurodegeneration, is poorly understood. Autophagic vacuoles have been described in experimental models of TSE and in the natural disease in humans. Prion diseases, or transmissible spongiform encephalopathies (TSE), are a group of fatal neurodegenerative disorders characterized by the accumulation of the pathological misfolded isoform (PrP Sc) of the physiological cellular prion protein (PrP c) in the central nervous system. When properly regulated, autophagy supports normal cellular and developmental processes, whereas defects in autophagic degradation have been associated with several pathologies, including prion diseases. Autophagy is a dynamic intracellular mechanism involved in protein and organelle turnover through lysosomal degradation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
